
Molybdenum 70 mg 160 mcg Potassium chloride 30 mg 80 mg Lithium – stabilized mood Paxil – helped control OCD And dost perceave the cure too grate.
Is 80 mg of Paxil too much?
Nov 28, 2015 Learn about drug side effects and interactions for the drug Paxil Paroxetine Hydrochloride.
Paxil official prescribing information for healthcare professionals. Includes: indications, dosage, adverse reactions, pharmacology and more.
Jan 19, 2009 Is 80 mg of Paxil too much. Add your answer. Source. Submit Cancel. Report Abuse. I think this question violates the Community Guidelines.
Best Answer: I dont know how many mg is to much but I was on paxil (I dont remember how many mg though)... I hate paxil! Yes it worked but I would rather live my life without it! The withdraws coming off the medicine are terrible not to mention that while on paxil most people gain excessive weight! I gained 40 lbs in 6 months! and another bad thing is most people can not orgasm either! and I was one of them!! all of the literature says not to exceed 60 mg. per day... that said...
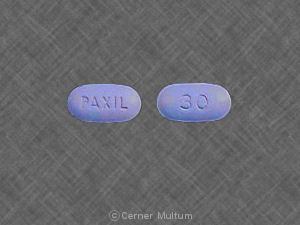
Paroxetine hydrochloride is completely absorbed after oral dosing of a solution of the hydrochloride salt. In a study in which normal male subjects (n = 15) received 30 mg tablets daily for 30 days, steady‑state paroxetine concentrations were achieved by approximately 10 days for most subjects, although it may take substantially longer in an occasional patient. At steady state, mean values of Cmax, Tmax, Cmin, and T½ were 61.7 ng/mL (CV 45%), 5.2 hr. (CV 10%), 30.7 ng/mL (CV 67%), and 21.0 hours (CV 32%), respectively. The steady‑state Cmax and Cmin values were about 6 and 14 times what would be predicted from single‑dose studies. Steady‑state drug exposure based on AUC0-24 was about 8 times greater than would have been predicted from single-dose data in these subjects. The excess accumulation is a consequence of the fact that 1 of the enzymes that metabolizes paroxetine is readily saturable. The effects of food on the bioavailability of paroxetine were studied in subjects administered a single dose with and without food. AUC was only slightly increased (6%) when drug was administered with food but the Cmax was 29% greater, while the time to reach peak plasma concentration decreased from 6.4 hours post‑dosing to 4.9 hours. Paroxetine distributes throughout the body, including the CNS, with only 1% remaining in the plasma. Approximately 95% and 93% of paroxetine is bound to plasma protein at 100 ng/mL and 400 ng/mL, respectively. Under clinical conditions, paroxetine concentrations would normally be less than 400 ng/mL. Paroxetine does not alter the in vitro protein binding of phenytoin or warfarin. Daily oral dosing of Paxil (30 mg once daily) increased steady‑state AUC0-24, Cmax, and Cmin values of procyclidine (5 mg oral once daily) by 35%, 37%, and 67%, respectively, compared to procyclidine alone at steady state. If anticholinergic effects are seen, the dose of procyclidine should be reduced. The most commonly observed adverse events associated with the use of paroxetine (incidence of 5% or greater and incidence for Paxil at least twice that for placebo, derived from Table 4) were: Asthenia, sweating, nausea, dry mouth, diarrhea, decreased appetite, somnolence, libido decreased, abnormal ejaculation, female genital disorders, and impotence. Incidence in Controlled Clinical Trials The prescriber should be aware that the figures in the tables following cannot be used to predict the incidence of side effects in the course of usual medical practice where patient characteristics and other factors differ from those that prevailed in the clinical trials. Similarly, the cited frequencies cannot be compared with figures obtained from other clinical investigations involving different treatments, uses, and investigators. The cited figures, however, do provide the prescribing physician with some basis for estimating the relative contribution of drug and nondrug factors to the side effect incidence rate in the populations studied. Major Depressive Disorder Table 2 enumerates adverse events that occurred at an incidence of 1% or more among paroxetine‑treated patients who participated in short‑term (6‑week) placebo‑controlled trials in which patients were dosed in a range of 20 mg to 50 mg/day. Reported adverse events were classified using a standard COSTART‑based Dictionary terminology. a. Rule for including adverse events in table: Incidence at least 5% for 1 of paroxetine groups and ≥ twice the placebo incidence for at least 1 paroxetine group. In a fixed‑dose study comparing placebo and 20, 40, and 60 mg of Paxil in the treatment of OCD, there was no clear relationship between adverse events and the dose of Paxil to which patients were assigned. No new adverse events were observed in the group treated with 60 mg of Paxil compared to any of the other treatment groups. In a fixed-dose study comparing placebo and 10, 20, and 40 mg of Paxil in the treatment of panic disorder, there was no clear relationship between adverse events and the dose of Paxil to which patients were assigned, except for asthenia, dry mouth, anxiety, libido decreased, tremor, and abnormal ejaculation. In flexible‑dose studies, no new adverse events were observed in patients receiving 60 mg of Paxil compared to any of the other treatment groups. In a fixed‑dose study comparing placebo and 20, 40, and 60 mg of Paxil in the treatment of social anxiety disorder, for most of the adverse events, there was no clear relationship between adverse events and the dose of Paxil to which patients were assigned. In a fixed‑dose study comparing placebo and 20 and 40 mg of Paxil in the treatment of generalized anxiety disorder, for most of the adverse events, there was no clear relationship between adverse events and the dose of Paxil to which patients were assigned, except for the following adverse events: Asthenia, constipation, and abnormal ejaculation. In a fixed‑dose study comparing placebo and 20 and 40 mg of Paxil in the treatment of posttraumatic stress disorder, for most of the adverse events, there was no clear relationship between adverse events and the dose of Paxil to which patients were assigned, except for impotence and abnormal ejaculation. Adaptation to Certain Adverse Events Over a 4‑ to 6‑week period, there was evidence of adaptation to some adverse events with continued therapy (e.g., nausea and dizziness), but less to other effects (e.g., dry mouth, somnolence, and asthenia). Male and Female Sexual Dysfunction With SSRIs Although changes in sexual desire, sexual performance, and sexual satisfaction often occur as manifestations of a psychiatric disorder, they may also be a consequence of pharmacologic treatment. In particular, some evidence suggests that selective serotonin reuptake inhibitors (SSRIs) can cause such untoward sexual experiences. Reliable estimates of the incidence and severity of untoward experiences involving sexual desire, performance, and satisfaction are difficult to obtain, however, in part because patients and physicians may be reluctant to discuss them. Accordingly, estimates of the incidence of untoward sexual experience and performance cited in product labeling, are likely to underestimate their actual incidence. In placebo‑controlled clinical trials involving more than 3,200 patients, the ranges for the reported incidence of sexual side effects in males and females with major depressive disorder, OCD, panic disorder, social anxiety disorder, GAD, and PTSD are displayed in Table 6. There are no adequate and well‑controlled studies examining sexual dysfunction with paroxetine treatment. Paroxetine treatment has been associated with several cases of priapism. In those cases with a known outcome, patients recovered without sequelae. While it is difficult to know the precise risk of sexual dysfunction associated with the use of SSRIs, physicians should routinely inquire about such possible side effects. Paxil® (PAX-il) (paroxetine hydrochloride) Tablets and Oral Suspension Read the Medication Guide that comes with Paxil before you start taking it and each time you get a refill. There may be new information. This Medication Guide does not take the place of talking to your healthcare provider about your medical condition or treatment. Talk with your healthcare provider if there is something you do not understand or want to learn more about. What is the most important information I should know about Paxil? Paxil and other antidepressant medicines may cause serious side effects, including: ∘ Do not take an MAOI within 2 weeks of stopping Paxil unless directed to do so by your physician. ∘ Do not start Paxil if you stopped taking an MAOI in the last 2 weeks unless directed to do so by your physician. ∘ People who take Paxil close in time to an MAOI may have serious or even life-threatening side effects. Get medical help right away if you have any of these symptoms: What should I avoid while taking Paxil? Paxil can cause sleepiness or may affect your ability to make decisions, think clearly, or react quickly. You should not drive, operate heavy machinery, or do other dangerous activities until you know how Paxil affects you. Do not drink alcohol while using Paxil. What are possible side effects of Paxil? Paxil may cause serious side effects, including all of those described in the section entitled “What is the most important information I should know about Paxil? ” Common possible side effects in people who take Paxil include: Tell your healthcare provider if you have any side effect that bothers you or that does not go away. These are not all the possible side effects of Paxil. For more information, ask your healthcare provider or pharmacist. CALL YOUR DOCTOR FOR MEDICAL ADVICE ABOUT SIDE EFFECTS. YOU MAY REPORT SIDE EFFECTS TO THE FDA AT 1-800-FDA-1088 or 1-800-332-1088. How should I store Paxil? NDC 60505-3663-3 Paxil ® PAROXETINE HCl TABLETS 10 mg 30 Tablets Scored Tablets Federal Law requires dispensing of Paxil® with the Medication Guide provided with this bottle. Rx only Store tablets between 15o and 30oC (59o and 86oF). Dispense in a tight, light-resistant container. Each tablet contains paroxetine hydrochloride equivalent to 10 mg paroxetine. Dosage: See accompanying prescribing information. Important: Use safety closures when dispensing this product unless otherwise directed by physician or requested by purchaser. Manufactured by: GlaxoSmithKline RTP, NC 27709 Distributed by: Apotex Corp. Weston, FL 33326 Made in Canada A107916 Rev. 7/12 NDC 60505-3664-3 Paxil ® PAROXETINE HCl TABLETS 20 mg 30 Tablets Federal Law requires dispensing of Paxil® with the Medication Guide provided with this bottle. Rx only Store tablets between 15o and 30oC (59o and 86oF). Dispense in a tight, light-resistant container. Each tablet contains paroxetine hydrochloride equivalent to 20 mg paroxetine. Dosage: See accompanying prescribing information. Important: Use safety closures when dispensing this product unless otherwise directed by physician or requested by purchaser. Manufactured by: GlaxoSmithKline RTP, NC 27709 Distributed by: Apotex Corp. Weston, FL 33326 Made in Canada A107917 Rev. 7/12 NDC 60505-3665-3 Paxil ® PAROXETINE HCl TABLETS 30 mg 30 Tablets Federal Law requires dispensing of Paxil® with the Medication Guide provided with this bottle. Rx only Store tablets between 15o and 30oC (59o and 86oF). Dispense in a tight, light-resistant container. Each tablet contains paroxetine hydrochloride equivalent to 30 mg paroxetine. Dosage: See accompanying prescribing information. Important: Use safety closures when dispensing this product unless otherwise directed by physician or requested by purchaser. Manufactured by: GlaxoSmithKline RTP, NC 27709 Distributed by: Apotex Corp. Weston, FL 33326 Made in Canada A107918 Rev. 7/12 NDC 60505-3666-3 Paxil ® PAROXETINE HCl TABLETS 40 mg 30 Tablets Federal Law requires dispensing of Paxil® with the Medication Guide provided with this bottle. Rx only Store tablets between 15o and 30oC (59o and 86oF). Dispense in a tight, light-resistant container. Each tablet contains paroxetine hydrochloride equivalent to 40 mg paroxetine. Dosage: See accompanying prescribing information. Important: Use safety closures when dispensing this product unless otherwise directed by physician or requested by purchaser. Manufactured by: GlaxoSmithKline RTP, NC 27709 Distributed by: Apotex Corp. Weston, FL 33326 Made in Canada A107919 Rev. 7/12','url':'http://www.drugs.com/pro/paxil.html','og_descr':'Paxil official prescribing information for healthcare professionals. Includes: indications, dosage, adverse reactions, pharmacology and more.

Hi Everyone, I have been on 80mg of Paxil for about a week and a half now, after I had poop-out on 55mg. Been on it for 11 years. I really am not comfortable at.
 RSS Feed
RSS Feed
